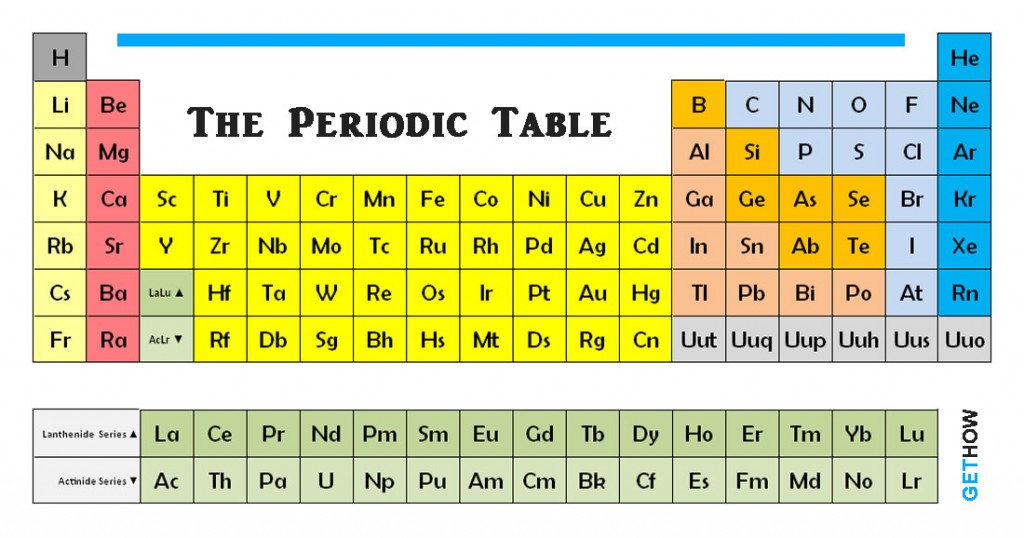
The lightest elements are hydrogen and helium, both created by Big Bang nucleosynthesis during the first 20 minutes of the universe in a ratio of around 3:1 by mass (approximately 12:1 by number of atoms). In October 2006, the synthesis of element 118 was reported however, element 117 has not yet been created in the laboratory. In 1955, element 101 was discovered and named mendelevium in honor of Mendeleev, the first to arrange the elements in a periodic manner. The current definition also avoids some ambiguities due to isotopes and allotropes.īy 1919, there were seventy-two known elements. In 1913, Henry Moseley discovered that the physical basis of the atomic number of the atom was its nuclear charge, which eventually led to the current definition. Put another way, a chemical element cannot be transformed into other chemical elements by chemical processes. In 1869, in Mendeleev's famous periodic table, shown below, there were sixty-six elements.įrom Boyle until the early 20th century, an element was defined as a pure substance that cannot be decomposed into any simpler substance. By 1818, Jöns Jakob Berzelius had determined atomic weights for forty-five of the forty-nine accepted elements. The first modern list of chemical elements was given in Antoine Lavoisier's 1789 Elements of Chemistry, which contained thirty-three elements, including light and caloric. In 1661, Robert Boyle showed that there were more than just four classical elements as the ancients had assumed. In other words, in combustion the fatty earth burns away. A piece of wood, for example, according to Becher, is composed of ash and terra pinguis when the wood is burnt, the terra pinguis is released, leaving the ash. These three earths correspond with Geber’s three principles. 
In modification on the ideas of Paracelsus, he argued that the constituents of bodies are air, water, and three types of earth: terra fluida, the mercurial element, which contributes fluidity and volatility terra lapida, the solidifying element, which produces fusibility or the binding quality and terra pinguis, the fatty element, which gives material substance its oily and combustible qualities. In 1669, German physician and chemist Johann Becher published his Physica Subterranea. Smoke represented the volatility (the mercury principle), the heat-giving flames represented flammability (sulphur), and the remnant ash represented solidity (salt). Mercury included the cohesive principle, so that when it left in smoke the wood fell apart. Paracelsus saw these principles as fundamental, and justified them by recourse to the description of how wood burns in fire. In 1524, Swiss chemist Paracelsus adopted Aristotle’s four element theory, but reasoned that they appeared in bodies as Geber’s three principles. Shortly thereafter, this evolved into the Arabic concept of the three principles: sulphur giving flammability or combustion, mercury giving volatility and stability, and salt giving solidity. 790 Arabian chemist Jabir ibn-Hayyan (Geber) postulated that metals were formed out of two elements: sulphur, ‘the stone which burns’, which characterized the principle of combustibility, and mercury, which contained the idealized principle of metallic properties. Aristotle defined an element as:Įlement – one of those bodies into which other bodies can be decomposed and which itself is not capable of being divided into other. Īdding to the four elements of the Greek philosopher Empedocles, in about 350 BC, Aristotle also used the term "element" and conceived of a fifth element called " quintessence", which formed the heavens. Plato assumed that the minute particle of each element corresponded to one of the regular polyhedra: tetrahedron (fire), octahedron (air), icosahedron (water), and cube (earth). The term 'elements' ( stoicheia) was first used by the Greek philosopher Plato in about 360 BCE, in his dialogue Timaeus, which includes a discussion of the composition of inorganic and organic bodies and is a rudimentary treatise on chemistry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
